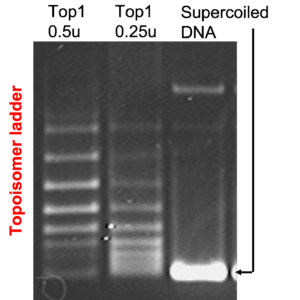
The purpose of the DNA Intercalator/Unwinding Kit is to determine if a compound induces DNA unwinding characteristic of DNA intercalators. Sufficient materials are provided for at least 50 independent determinations.
Using this kit to detect a DNA intercalator (DI): Increasing DI in the presence of topoisomerase I results in an increase in reference DNA supercoiling (as shown above).
DNA Unwinding Kit Contents
-Substrate DNA: pHOT1 DNA
-Relaxed DNA Marker: 0.05 ug/ul in 1x gel loading buffer
-10 x Topo I assay buffer,
-10x Gel Loading buffer
-10% SDS
-mAMSA Stock, Lyophilized (control intercalator)
–Detailed Instruction Manual
The following materials and reagents are not supplied with this kit but will be required:
Agarose and related electrophoretic supplies and equipment
Transilluminator and photodocumentation system
DNA extraction and purification related reagents: Proteinase K, Choloroform: Isolamyl alcohol (24:1)
The kit can be shipped at ambient temperature. Store at 4°C.
TG1025 Kit Protocol
TG1025 Material Safety Data
The DNA Unwinding Kit Theory
Unwinding of the double strands of the DNA helix is a hallmark feature of intercalating drugs, such as chloroquine, ethidium bromide and m-AMSA. As part of characterization of novel agents, it is often important to know whether a novel compound is a DNA intercalator. This can easily be tested by examining the effect of the agent on the average linking number of topoisomers. Specifically, the influence of a compound on the distribution of topoisomers (generated by relaxation of closed circular DNA with topo I) will reveal the presence of a DNA intercalator. For example, intercalators induce a change in the average DNA linking number of a population of topoisomers generated by topo I (compared to reactions lacking the test compound). Stated differently, DNA intercalation induces unwinding of the DNA template and thereby alters a parameter called Twist (Tw). Defining the details of DNA supercoiling is beyond the scope of this introduction; however, excellent reviews on the subject can be found in the literature1-4). Briefly, the relation between strand twists, linking and writhe can be expressed by the simple equation:
- Lk = Tw + Wr
- Lk is linking number: An integer describing the number of times one strand is linked with the other
- Tw is the arrangement in space of one strand twisting about the other and is continuously variable
Wr is the writhing number which describes deformation of the helical axis in 3D space. Wr is also a continuously variable parameter. Lk is a fixed property of a plasmid and cannot change no matter how Tw and Wr fluctuate; Lk can only change by breaking/resealing and subsequent strand passing (topoisomerase I does this for example). By extension, when a DNA molecule if fully relaxed by topo I, Wr approaches zero and Lk = Tw. For relaxed B form DNA, Lk equals the number of base pairs in the plasmid divided by 10 (since each strand of the duplex makes a full 360o rotation every 10 bp or so). The products of relaxation by topo I strongly influenced by conditions that are extant at the time of strand closure; as a population of DNAs one sees a gaussian distribution of topoisomers (see below). Each topoisomer will differ from its nearest neighbor by a Lk difference of one. DNA intercalators are known to locally unwind the DNA (5). Upon intercalation, Tw is reduced and since topo I can sense this change and adjust Lk, the equilibrium distribution of topoisomers is subsequently adjusted; therefore, the Lk now reflects the Tw parameter of the intercalated form of the DNA. Upon terminating the reaction with SDS, digesting with proteinase and extracting the DNA with organic solvents, protein and drug are removed and any alteration in the population distribition of topoisomers is revealed by running an agarose gel. The gels are run to high resolution to display topoisomers; thus, gels are generally run without ethidium bromide at a low voltage for 12 hrs. or more. These DNA unwinding experiments are informative and reveal useful and subtle information about drug/DNA interaction. Comparison of DNA unwinding with well known DNA intercalators (such as m-AMSA) give the investigator some idea of the potency of DNA binding. One important caveat, however is that the investigator must be sure that the test drug is not inhibiting topo I activity. Proper controls will eliminate this potential complication.