Academic pricing is shown. For-profit institutions should contact TopoGEN directly for a quote.
Cell Based Screening Kit for Homologous Repair (HR) using CRISPR-Cas9
DNA Repair pathways in animal cells can be divided into two main categories: HR and NHEJ. HR or homologous recombination is a minor pathway but very important in protecting cells from genotoxicity. The process has two key requirements as well: a homologous sequence, usually available after DNA replication when the genome is 4N, and S-phase. A specific reporter based assay for HR can be very beneficial for anti-cancer drug discovery projects, learning more about the process of DNA HR repair and establishing intersecting pathways and druggable pathway targets.
This is a cell-based reporter kit designed to allow the customer to screen or identify agents (drugs, natural products, small molecules, synthetics, miRNAs, and genes) that affect or impact the process of HR DNA repair. The kit uses GFP as an in vivo readout for the HR pathway and Crispr-CAS gRNA plasmid that drives a DS DNA break in Cassette 1. This DS break induces HR in a mutated GFP reporter gene, which recombines with Cassette 2 containing WT GFP leading to GFP+ cells after completing HR.
DNA is continually being exposed to genotoxic agents leading to cell death and/or changes in gene expression. Of the various forms of DNA damage, the most dangerous are DNA double-strand breaks (DSBs), which may create serious problems arising from inappropriate recombination such as chromosomal translocations. To deal with the threats posed by DSBs, cells have developed multiple mechanisms to detect, signal, and repair the regions in chromatin. Two main pathways, homologous recombination (HR) and non-homologous end-joining (NHEJ), are involved in the repair of DSB. These pathways are further subdivided into more specific sub-pathway processes. In prokaryotes, HR has been known to be a major pathway for the repair of DSBs, while in eukaryotes, NHEJ was thought to be preferred. More recently, HR has also been shown to be a operate in mammals. These pathways are largely distinct from one another and function in complementary ways. NHEJ involves the ligation of two DNA ends without homology and tends to be error prone while HR is high fidelity and essentially error free. In the HR process (sometimes referred to as gene conversion), a donor DNA sequence with homology to both sides of the DSB supplies genetic information to repair the DSB. The homologous sequence is copied into the broken locus, making the repaired locus an exact copy of donor sequence, without altering the donor sequence (Fig. 1).
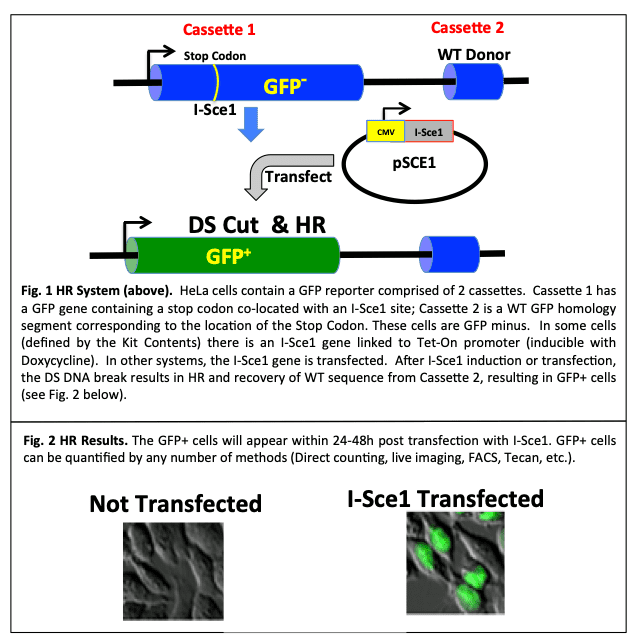
A cell based/cell context system has been designed to allow researchers to examine and interrogate the HR process in live HeLa cells. The HR Kit uses a twin GFP cassette that converts from GFP negative to GFP positive cells using homologous recombination (HR). DNA repair via HR (as gene conversion) will result since a wild type (homologous) GFP segment is present in close proximity to the DNA break. The DS break initiates HR and using the WT sequence as a homology template (located in Cassette 2) the gene converts to WT and GFP positive cells appear (Fig. 1). HR is triggered by a DS break which is achieved using a gRNA with CRISPR-CAS. The Cas9-GFP-gRNA-mCherry plasmid provides an internal transfection control (Red+ cells correspond to the transfection efficiency). Cells are that are dually labeled (Red+/GFP+) have completed HR while cells expressing only GFP+ correspond to background cells that initiated HR or simply converted to WT GFP status by other mechanisms. Table 1 is a summary of the various readouts and interpretation of results.
| Cell Signal: | Readout: | INTERPRETATION | % Observed |
|---|---|---|---|
| No Flourescence | None | No HR and no mCherry Transfected | Not Done |
| GFP+ Only | Green Cells but no Red | Background GFP+ cells; not Induced HR | 0.5-0.8% |
| mCherry+ Only | Red Cells but no Green | Cells Transfected with mCherry gRNA that have not initiated HR (HR is cell cycle dependent and cells must be in S-phase) | 50% (variable, based on efficiency) |
| GFP+ mCherry+ | Green and Red Cells | Cells Transfected with Cas9-gRNA, DS Break Induced in Cassette 1, HR Completed | 8-10% |

