This kit contains all the reagents for routine detection of covalent topoisomerase/DNA intermediates. The DNA target that is included that is nonradioactive and a protocol is included for end labeling. All other reagents have been pretested and are certified to function in the SDS-K+ precipitation method based upon the analytical method described by Trask et al. (EMBO J. 3: 671-676, 1984). Note that this kit is designed for in vitro studies; however, we also have an in vivo kit to analyze endogenous topo I and II binding to the genome. See 1011-1 for details.
Kit Contents:
-Substrate DNA: Lambda DNA (ready for labeling).
-10 x Topo I assay buffer
-10 x Topo II assay buffer
-10% SDS
-10X TKE wash buffer
-2.5M KCl
-Buffer A1 reaction buffer
A Detailed Manual that is divided into three major sections:
-Procedure for End Labeling of the DNA Substrate
-Procedure for the SDS-K+ Precipitation
-Controls
This TopoGEN kit is designed to quantify formation of the covalent protein/DNA intermediate that forms when topoisomerase I or II reacts with DNA. Detecting the complex is greatly enhanced by topo poisons that stabilize the reaction (covalent) intermediate. Since a radioactive DNA substrate is used, the procedure is quantitative and can be used to compare or identify different inhibitors. Radioactivity is not supplied; however, detailed instructions are included which describe the procedure (along with an appropriate DNA target).
The following materials and reagents are not supplied with this kit but will be required:
95% and 70% Ethanol
Glass Fiber Filters (used for TCA precipitations, such as Whatman GF/C)
Filtration manifold or single Millipore Filter stack
alpha-[32P]-dATP or alpha-[32P]-dTTP
E. coli DNA polymerase (Klenow) and EcoR1
10X EcoRI digestion buffer
Deoxynucleoside triphosphates
Spin Columns (optional)
Scintillation vials, Scintillation fluid
Background:
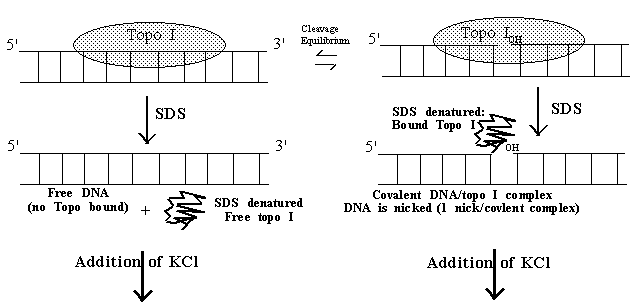
Both type I and II topoisomerases can be trapped in a covalent complex with DNA by adding protein denaturants (alkali, acid or detergents) while the enzyme is actively engaged in breaking and resealing steps on DNA. Although the detailed mechanism of trapping by denaturation is not known, it is generally accepted that capturing the covalent intermediate or cleavable complex is difficult because the intermediate has a relatively short life time. Thus, with normal amounts of topoisomerase I or II (i.e., sufficient enzyme to observe catalysis), detecting the covalent complex by denaturation requires the presence of topo inhibitors which tend to extend the life time of the cleavage complex. (Strictly speaking, these types of inhibitors are in fact better described as poisons.) Formation of a covalent intermediate can be exploited as a means of identifying novel topo poison since the complex is a functional intermediate of the catalytic cycle of topo. Covalent topo/DNA complexes are generated by adding SDS to a reacon in which the enzyme is nicking and resealing DNA; a fraction (typically <5%) of the topo is trapped in a covalent complex with DNA. SDS binds only protein (not DNA) and non-covalently bound topo is irreversibly inactivated. Upon addition of KCl, insoluble crystals of potassium dodecyl sulfate form (we refer to this as SDS-K+) which co-precipitate free protein and only those DNAs covalently bound to topo I. If the DNA is labeled, the amount of label in the precipitate is a measure of DNA molecules with covalently bound (denatured) topo. DNA in the SDS-K+ precipitate can be recovered and shown to be all nicked (topo I reactions) or a mixture of nicks and double strand breaks (topo II), as expected from what we know of the reaction mechanism. The SDS-K+ method is particularly useful for quantifying topo DNA covalent complexes to: 1) identify topo I or II inhibitors or poisons and 2) to facilitate comparison of different inhibitors in their ability to increase the life time of the cleavage complex.
Count Precipitate Count Precipitate
The Buffers and DNAs should be stored at 4°C for the short term and at -20°C for the long term.

